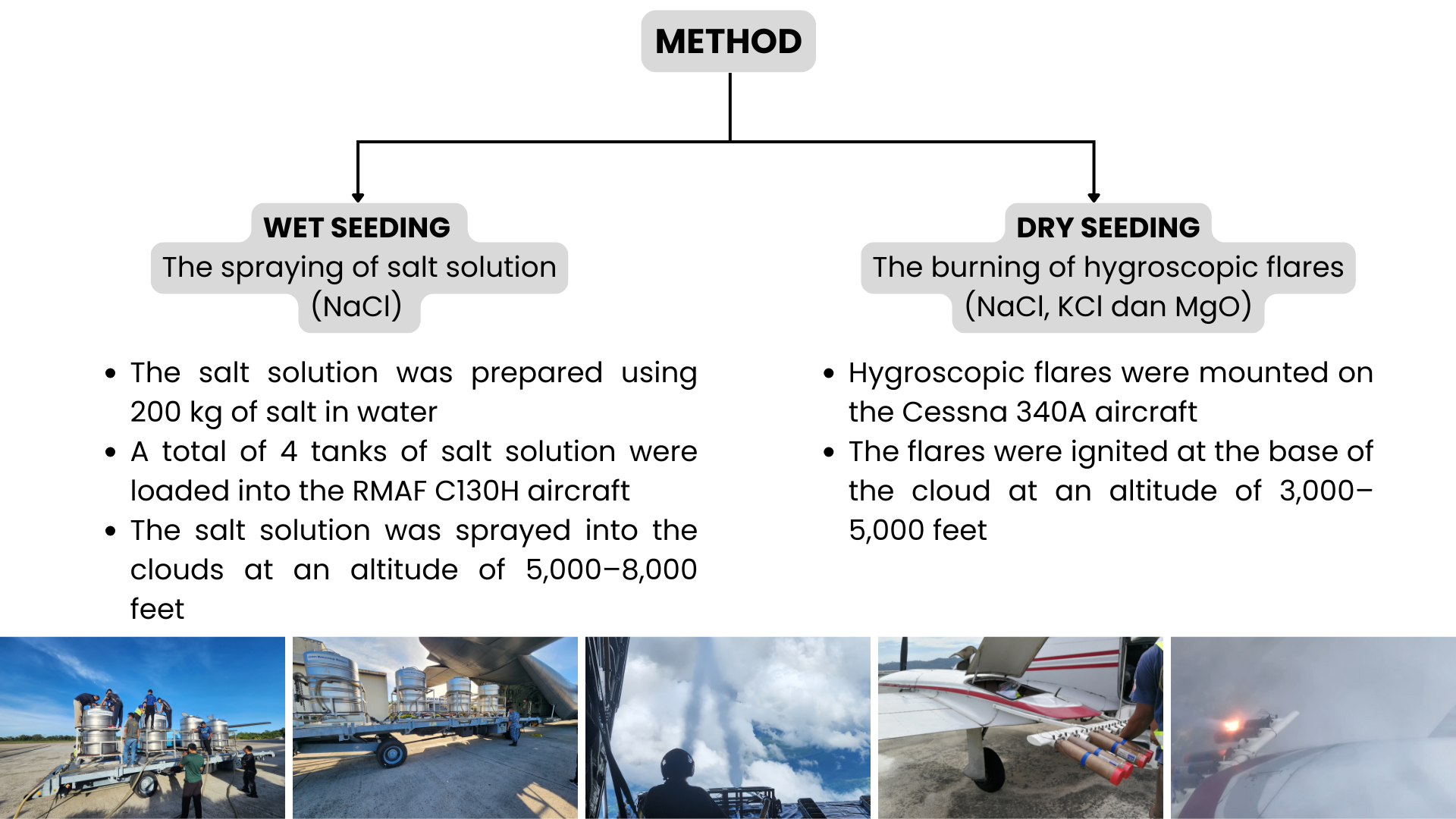
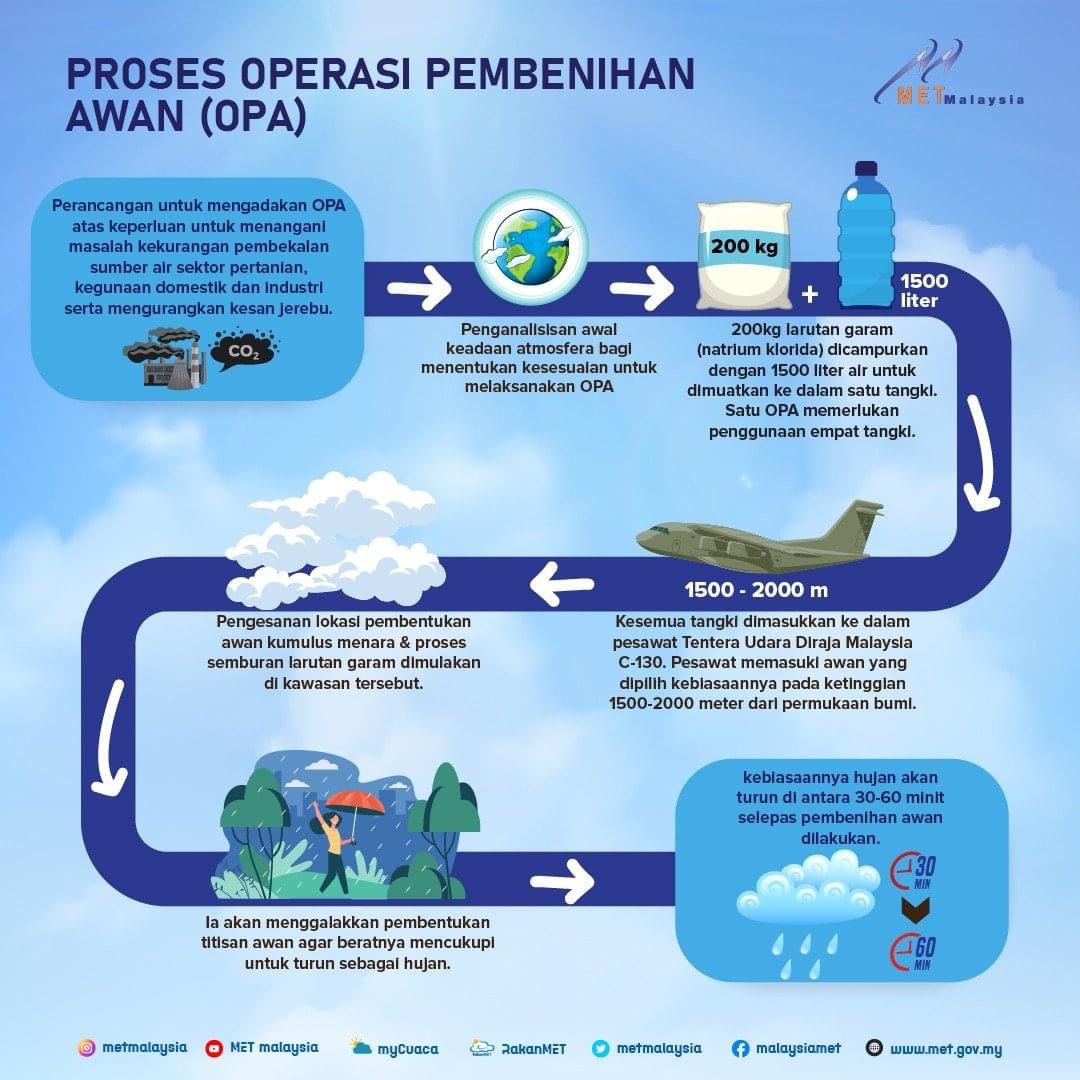
- Home
- Corporate Profile
- Forecast
- Earthquake
- Observation
- Climate
- Publication
- Education
- Contact Us
-
Talian Hotline1-300-22-1638


OFFICIAL WEBSITE OF
MALAYSIAN METEOROLOGICAL DEPARTMENT
MINISTRY OF NATURAL RESOURCES AND ENVIRONMENTAL SUSTAINABILITY
Talian Hotline
1-300-22-1638